The Linac-MR Project




The Whole Body Linac MR (Linac MRI) Radiotherapy Hybrid that Could Significantly Improve Cancer Patient Survival Rates
Overview
Treating tumours with radiation has long been an accepted technique for treating cancer. However, targeting tumours in organs that may move such as lung, prostate, and stomach can be a challenge. Also sparing radiation sensitive organs such as the rectum and bladder which are adjacent to a tumour site provides an additional complication. To meet this challenge, the Linac MR Research group at the Cross Cancer Institute(CCI) has built the first functioning Linac-MR system that merges the imaging capabilities of a Magnetic Resonance Imaging (MRI or MR) system with the radiation treatment capabilities of a Linear Accelerator (linac).
A Hybrid Linac MRI System
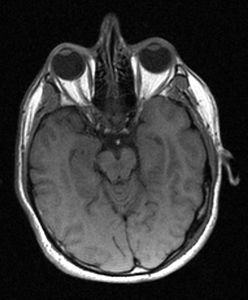
The first whole body clinical linac MRI hybrid (linac-MR) began installation in 2013 at the CCI, Edmonton AB, Canada. The linac-MR integrates into one machine, a linac and an MRI. Installation of the system in a pre-existing radiotherapy vault (6 m x 6m) was performed without the need to remove walls or ceilings. Acceptance testing of the MR component is now complete, and radiation testing is progressing. The world's first images from a linac-MR on a human volunteer were obtained in July 2014.
The linac-MR consists of an isocentrically mounted 6 MV linac that rotates in-unison with a biplanar 0.5 T MRI in transverse plane. The MRI's Bo field and the central axis of the 6 MV beam are parallel to each other. The optimized fringe field results in insignificant increase in entrance dose. This parallel configuration avoids large increases in dose at tissue/air interfaces and at beam exit due to electron return effect that occurs in the perpendicular configuration(Bo field is perpendicular to the central axis). The open configuration of the systems allows real-time MRI guided radiotherapy of all tumors including peripheral tumours (eg, in breast, etc) with imaging and treatment performed concurrently.
Our group at the Cross Cancer Institute were first to demonstrate concurrent MR imaging and linac-irradiation of head-size phantoms in 2008, both systems installed on a single gantry. The prototype and design solutions have been described in our 60+ peer-reviewed articles. The current functional whole-body rotating linac-MR system is designed and built based on the engineering physics solutions given in the publications and raw-data from the first prototype.
The current system is mechanically well balanced and rotates at 1 rpm using only a 1 HP motor. The 3D magnetic field mapping demonstrates minimal perturbation in magnetic field homogeneity with gantry rotation which is easily and effectively shimmed by gradient coils alone. The Larmor Frequency varies with gantry angle due to the Bo interaction with room shielding and to the directional changes of the Earth's magnetic relative to Bo, and this change closely follows our predictions calculated previously. Angle dependent 3D magnetic field maps and Larmor Frequency are used to automatically and optimally create image acquisition parameters for any gantry angle.
Metrics calculated from images obtained at different rotating angles, and various acquisition speeds from a human volunteer, and ACR and other phantoms, show that the image quality is comparable to those of clinical MRI systems, and thus satisfy the requirements for real-time MR-guided radiotherapy.